Background: Myelodysplastic syndrome (MDS) is a clonal hematopoietic neoplasm that results in bone marrow failure and frequently leads to acute myeloid leukemia (AML). Hypomethylating agents (HMA) are the only FDA-approved treatment for MDS and among few options for chemotherapy-ineligible patients with AML. There are limited options for patients in whom HMA therapy fails and who are not candidates for allogeneic stem cell transplant, so therapies that complement or restore sensitivity to HMAs are needed. Studies have shown increases in bone marrow microvessel density (MVD) and angiogenic markers in patients with MDS and AML. A decrease in MVD has been shown to correlate with response to hypomethylating agents in MDS. A receptor-ligand interaction, comprised of receptor EphB4 and membrane localized ligand EphrinB2, mediates angiogenesis in normal tissue and appears to be a target unique to many cancer types. We have previously shown EphB4 to be highly expressed and a driver of leukemic cell survival in a subset of AML patients. Our group has developed a human fusion protein, sEphB4-HSA, that blocks bidirectional signaling induced by EphB4-EphrinB2 interaction to inhibit tumor cell proliferation and angiogenesis. In phase I clinical trials of sEphB4-HSA in various tumor types, there were no myelosuppressive effects and minimal toxicity. Given its safety in phase I and potential to inhibit leukemic cell proliferation and angiogenesis, we proposed a pilot trial to evaluate the safety of sEphB4-HSA in combination with HMAs in MDS and AML patients who have failed treatment with HMAs.
Methods: This pilot study was designed to enroll 6 patients with relapsed/refractory intermediate or high-risk MDS and 6 patients with AML refractory to or relapsed to HMA treatment and who are deemed unfit for chemotherapy. Treatment consisted of sEphB4-HSA 15 mg/kg IV every 2 weeks in combination with the FDA-approved HMA most recently or currently being used for treatment (decitabine 20mg/m2 IV/1hr on days 1 to 5 every 28 days or azacitidine 75mg/m2 SC or IV on days 1 to 7 every 28 days). Patients were treated for as long as they were receiving clinical benefit up to 12 months. The primary endpoint was toxicity and tolerability of sEphB4-HSA in combination with HMA. Toxicity was assessed and graded after each cycle according to the CTCAE version 4. Tolerability was defined as the ability to complete two cycles of treatment without the occurrence of dose-limiting toxicity. A secondary efficacy endpoint was to assess best overall response, based on the IWG Working Group Criteria for MDS and AML, during the first two cycles of treatment. Enrollment was stopped after 7 patients due to expiration of funding.
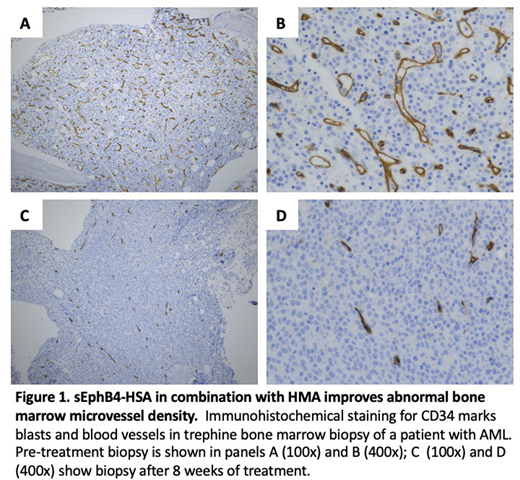
Results: Three patients with intermediate-risk MDS were treated for a median duration of 6 cycles (2-12) and 4 patients with AML were treated for 2 cycles. Median age was 75.5 years (67.9-84.8) and 57.1% were male. HMA included azacitadine in 6 patients and decitabine in one patient. There were no dose-limiting toxicities. There were 8 grade 3/4 events attributed to HMA, which included: neutropenia (2), thrombocytopenia (3), and leukopenia (3). There were 3 grade 3/4 events attributed to sEphB4, which included: febrile neutropenia (1), leukopenia (1), and hypertension (1). Of the MDS patients, 2 had stable disease, 1 patient after 2 cycles and 1 patient after 4 cycles. One patient achieved a hematologic improvement-erythroid after 6 cycles. AML patients had no disease response. Reasons for treatment discontinuation were death (1), disease progression (2), patient's decision (1), physician's decision (2), and hospice (1). Notably, a comparison of bone marrow biopsies at baseline and after 8 weeks of treatment demonstrated a decrease in MVD (Figure 1).
Discussion: This pilot study found sEphB4 in combination with HMAs to be tolerable with no significant toxicity beyond that expected with HMA therapy and associated with potential clinical benefit in MDS patients. Improvement in abnormal bone marrow MVD may indicate a potential for sEphB4-HSA plus HMA therapy to alter the malignant microenvironment in MDS/AML.
No relevant conflicts of interest to declare.
Author notes
Asterisk with author names denotes non-ASH members.

This feature is available to Subscribers Only
Sign In or Create an Account Close Modal